Properties
Key Properties
Silicon Dioxide is an odorless compound that is made when silicon is exposed to oxygen and it oxidizes to form
SiO2 or Silicon Dioxide, which is also known as Silica. It is a group IV metal
oxide and it is also an acidic oxide, which means that when it is in water it
produces hydrogen ions. It has good abraision resistance, good electrical
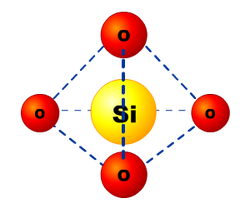
insulation, and high thermal stability. Silica is insoluble in all acids except Hydrofluoric acid (HF). In its crystalline form (quartz) the Si atom shows a tetrahedral formation with 4 oxygen atoms around an Si atom as seen in the picture (left).
Other Properties
Density
Crystalline Form (Quartz) ----------------------------------------------------- 2.65 g/cm^3
Fused Silica ---------------------------------------------------------------------- 2.2 g/cm^3
Molar Mass -------------------------------------------------------------------------- 60.0843 g
Melting Point ------------------------------------------------------------------------ 1710 degrees Celsius
Boiling Point ------------------------------------------------------------------------- 2230 degrees Celsius
Natural Abundance in Crust ----------------------------------------------------- 2.82x10^5 mg/kg
Natural Abundance in Sea ------------------------------------------------------- 2.2 mg/l
Silicon Dioxide is an odorless compound that is made when silicon is exposed to oxygen and it oxidizes to form
SiO2 or Silicon Dioxide, which is also known as Silica. It is a group IV metal
oxide and it is also an acidic oxide, which means that when it is in water it
produces hydrogen ions. It has good abraision resistance, good electrical
insulation, and high thermal stability. Silica is insoluble in all acids except Hydrofluoric acid (HF). In its crystalline form (quartz) the Si atom shows a tetrahedral formation with 4 oxygen atoms around an Si atom as seen in the picture (left).
Other Properties
Density
Crystalline Form (Quartz) ----------------------------------------------------- 2.65 g/cm^3
Fused Silica ---------------------------------------------------------------------- 2.2 g/cm^3
Molar Mass -------------------------------------------------------------------------- 60.0843 g
Melting Point ------------------------------------------------------------------------ 1710 degrees Celsius
Boiling Point ------------------------------------------------------------------------- 2230 degrees Celsius
Natural Abundance in Crust ----------------------------------------------------- 2.82x10^5 mg/kg
Natural Abundance in Sea ------------------------------------------------------- 2.2 mg/l
